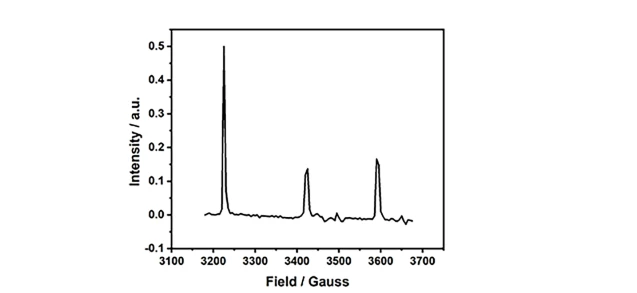
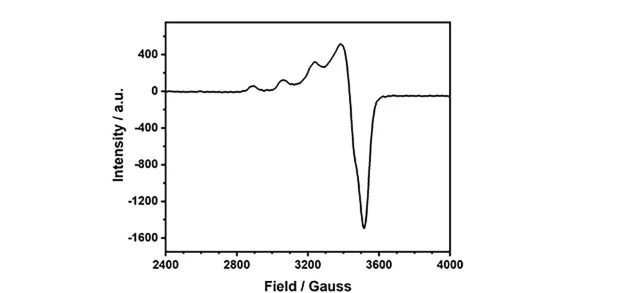
The CIQTEK EPR200-Plus is a floor-standing EPR spectrometer with enhanced sensitivity for robust continuous wave (CW) EPR measurements. The model can be customized with three different sizes of electromagnets.
An upgraded version, CIQTEK EPR300, is another floor-standing CW EPR spectroscopy with higher sensitivity and optional Q-band extension.
>> EPR200-Plus Accessories: Dual Mode Resonator, High-temperature System, Liquid Nitrogen Variable Temperature With Cryostat, Liquid helium Variable Temperature, Liquid Helium-free Dry Cryogenic System, Time-resolved EPR System, Goniometers, Irradiation system, Flat cell.
Electron paramagnetic resonance (EPR) or electron spin resonance (ESR) spectroscopy is a powerful analytical method to study the structure, dynamics, and spatial distribution of unpaired electronics in paramagnetic substances. It can provide in-situ and non-destructive information on electron spins, orbitals, and nuclei at the microscopic scale. EPR spectroscopy is particularly useful for studying metal complexes or free radicals so it has important applications in the fields of chemistry, materials, physics, environment, etc.